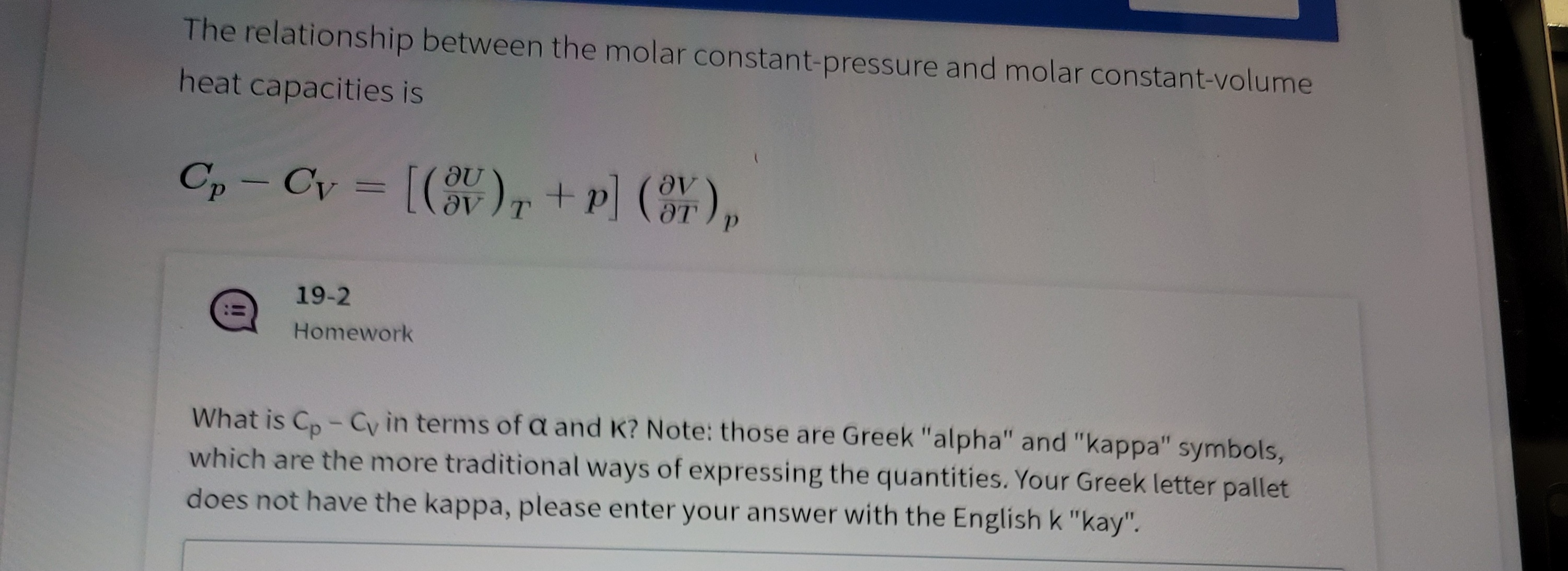
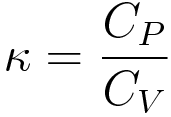Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download

Development in the Concept of Bacterial Polysaccharide Repeating Unit-Based Antibacterial Conjugate Vaccines | ACS Infectious Diseases
Ideal and Real Discharge Coefficients – Using Fundamental Equations of State in Mass-Flow Measurements with Sonic Nozzles R. S

OpenFOAM-9 multiphaseEulerFoam, wallBoiling, changing table Hs to accommodate new pressure : r/OpenFOAM

Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube

Biophysik Physikalische Chemie - Teil Physikalische Chemie a) Wie lautet der 1. Hauptsatz der - Studocu

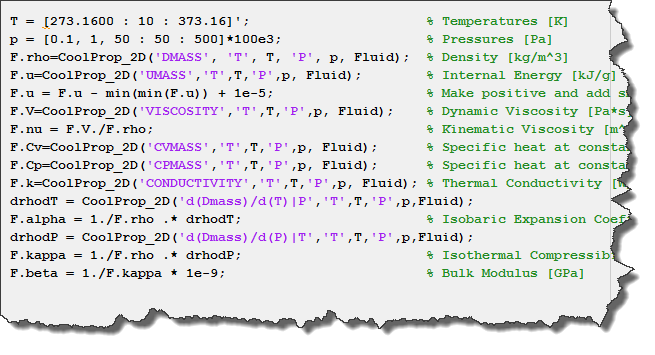









![Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub Question] Composition summation · Issue #347 · usnistgov/REFPROP-wrappers · GitHub](https://user-images.githubusercontent.com/58614082/103503111-e8fcdf00-4e18-11eb-99f3-f320c36dd40c.png)